This section is an adaptation of the paper: “DICER1: mutations, microRNAs and mechanisms”, published in Nature Reviews Cancer by Foulkes, Priest and Duchaine (PMID 25176334). For more information, please refer to the original article.
For an up to date list of papers published about the DICER1 gene and DICER1 syndrome, please click here.
Molecular Functions
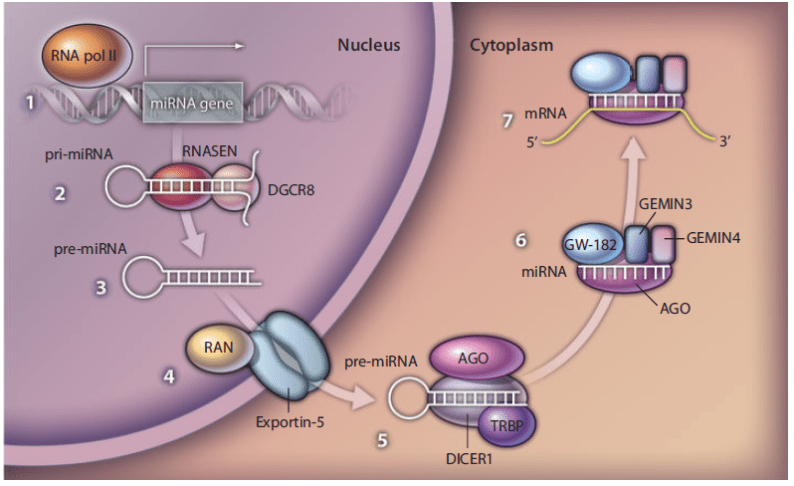
The Dicer protein is an endoribonuclease responsible for the production of mature microRNAs (miRNAs), which are small single-stranded RNA molecules that target messenger RNAs (mRNAs) and have an important role in regulating gene expression by repressing protein synthesis. The human form of the Dicer protein is called DICER1, encoded by the DICER1 gene, and is widely expressed across tissues. DICER1 is involved in the miRNA-mediated gene silencing pathway, where a miRNA imperfectly hybridizes to target mRNAs. This, in turn, generally leads to repression of mRNA translation and initiation of mRNA decay. A primary miRNA (pri-miRNA) transcript is produced via transcription of a miRNA gene by the RNA polymerase II enzyme. The pri-miRNA is then processed in the nucleus by DROSHA (also known as RNASEN), an RNA endonuclease, and the RNA binding protein DGCR8 (also known as PASHA), resulting in the production of an RNA stemloop that contains the miRNA. This premature miRNA (pre-miRNA) hairpin is subsequently exported into the cytoplasm through nuclear pores. The cytoplasmic DICER1 protein then cleaves the pre-miRNA to release the mature short (~21 nucleotide) miRNA. This miRNA is then loaded into an Argonaute (AGO) protein forming the core of the miRNA-induced silencing complex (miRISC). The miRISC then hybridizes to targeted mRNAs in order to engender their silencing.
DICER1 Structure
- The human DICER1 gene is located on chromosome 14q32.13 and is comprised of 27 exons with 1,922 amino acids. The DICER1 protein is a large multi-domain enzyme shaped like an ‘L’.
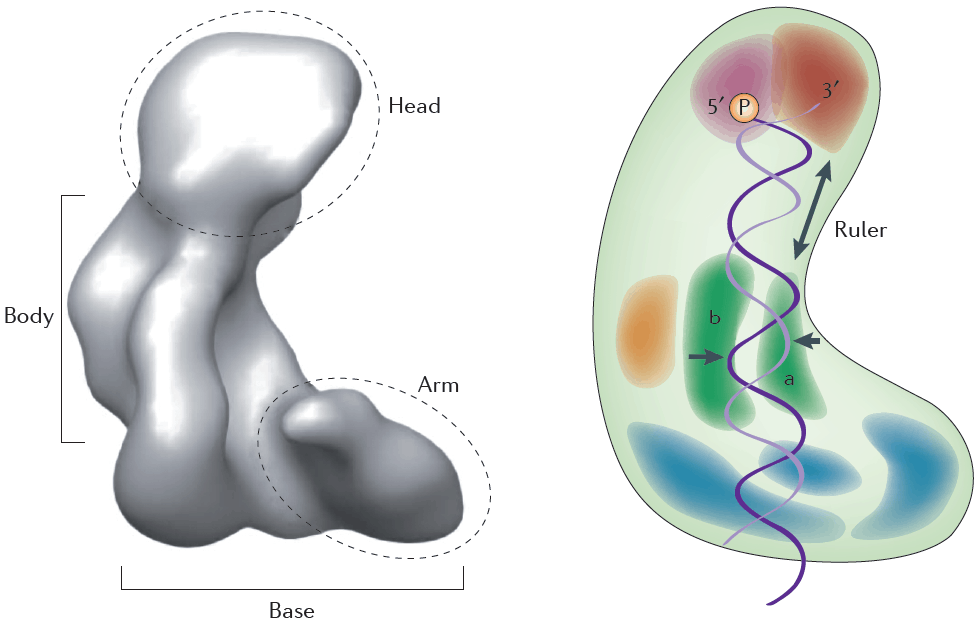
- At the top of the ‘L’ are the PAZ and Platform domains. These domains are binding pockets for double-stranded RNA (dsRNA). The 3’-overhang of the dsRNA substrate binds at the PAZ domain, and the 5’-overhang binds at the Platform domain.
- The lower half of DICER1 consists of the RNase IIIa and RNase IIIb domains, which dimerize to form the critical catalytic core of the enzyme and each cleave one strand of the dsRNA substrate. The RNase IIIa domain is responsible for producing 3p miRNAs from the 3’ strand, and the RNase IIIb domain produces 5p miRNAs from the 5’ strand. DICER1 requires magnesium and/or manganese ions in order to perform the catalytic cleavage. These metal ions are bound to ion-binding residues within the RNase III domains at amino acid residues E1320 and E1564 in the RNase IIIa domain, and D1709 and E1813 in the RNase IIIb domain.
- Clinical correlation: Most individuals with DICER1 syndrome-related tumors are found to harbour two mutations in DICER1. These typically consist of one heterozygous germline truncating (inactivating) mutation, which may be located at any position throughout the gene, coupled with a somatic (or tumor-specific) missense mutation, which consistently affect one of the metal-ion binding resides within the RNase IIIb domain or an adjacent residue. Almost all tumors and dysplasias associated with the syndrome have been found to harbour such ‘hotspot’ mutations. In a very small subgroup of patients with DICER1 syndrome, the initial mutational events are mosaic RNase IIIb mutations; the phenotype in these cases may be more severe
- The RNase III domains are separated from the dsRNA binding domains by a linker, which contains a connector helix. This holds the dsRNA phosphate backbone in place. It also acts as a ‘ruler’ by positioning the pre-miRNA along the enzyme so that the enzyme can cleave the dsRNA into miRNAs of appropriate sizes.
- At the base of the L is the DExD/H box helicase domain. This domain is thought to clamp down on the dsRNA substrate.
Clinical Features/Phenotype
In 2009, heterozygous germline DICER1 mutations were identified in 11 families with one or more individuals with pleuropulmonary blastoma (PPB) and family histories of various other tumors. In so doing, a novel hereditary cancer predisposition syndrome was described, entitled by Slade et al as “DICER1 syndrome” (OMIM #601200).
Since then, the phenotypic spectrum has been expanded through the identification of many other families with germline DICER1 mutations, with and without PPB, to include predisposition to additional tumors, notably cystic nephroma (CN), ovarian sex-cord stromal tumors (OSCSTs, especially Sertoli-Leydig cell tumors (SLCT)), multinodular goiter (MNG), cervical embryonal rhabdomyosarcoma (cERMS) and pituitary blastoma (PitB), among others. Bilateral involvement is not unusual in paired organs. DICER1 germline mutations have been described in individuals with various other tumor types, and new tumors are being added to the DICER1 syndrome spectrum. See the “Phenotypes” tab for more information regarding the tumors associated with DICER1 syndrome.
Many of the tumors observed in DICER1 syndrome are quite rare or unusual. Germline genetic testing and genetic counseling for a patient and/or family is suggested for either of the following circumstances: (a) a diagnosis of any one of the highly characteristic tumors (e.g. PPB, CN, PitB); (b) a co-occurrence in a patient or family of any of the DICER1-associated tumors.
Many of the DICER1-related tumors present before the age of approximately 15 to 25 years, with some presenting in very early childhood such as PPB, CN, and PitB. However, thyroid disease and some tumors (such as cERMS and OSCST) may appear as late as ages 40 to 50.
The penetrance of DICER1 mutations for tumor development is not definitively known at this time, but appears to be low and variable, even within families. Further studies are needed to clarify the true penetrance of DICER1 syndrome, as it can present with sub-clinical phenotypes such as lung or renal cysts, or miniscule thyroid nodules. Penetrance appears to be higher in females due to the occurrence of gynecological tumors and more frequent thyroid disease. It appears that at least 50% of female carriers and perhaps 80% of male carriers will remain clinically unaffected.
DICER1 Mutations – Germline and Somatic
DICER1 syndrome is characterized by heterozygous germline mutations (mutations in one of the two copies of an individual’s DICER1 gene in every cell of their body) and exhibits autosomal dominant inheritance. Children of an individual with a DICER1 germline mutation have a 50% chance of inheriting the mutation, and thus being at risk for DICER1-related tumors, and a 50% chance of not inheriting the mutation, and thus not being at increased risk for DICER1-related tumors. Most DICER1 mutations are inherited from a parent who also carries the mutation (who may or may not have a personal history of DICER1-related tumors). In about 20% of cases, family testing reveals that the DICER1 mutation is de novo in the affected individual. In this case, siblings of the affected individual are presumed not to be at risk for DICER1-related tumors. Gonadal mosaicism, the phenomenon in which a parent does not carry a germline DICER1 mutation but has a DICER1 mutation in a subpopulation of cells specifically localized to the gonads, has not been observed in DICER1 syndrome, but is theoretically possible. Therefore, genetic testing could be considered for siblings of children with an apparently de novo mutation, although a positive result is extremely unlikely.
Germline mutations in DICER1 are typically truncating (although pathogenic missense mutations have rarely been observed) and have been identified throughout the gene. Germline DICER1 mutations have been observed across many ethnic groups, and there have been no observed founder effects. There are no consistent genotype-phenotype correlations between germline mutations and associated tumor penetrance.
DICER1 syndrome fits the model of a tumor suppressor gene (TSG), in that the gene is functionally inactivated by biallelic mutations (one germline and one somatic mutation, in the context of DICER1 syndrome). However, DICER1 differs from most other TSGs in that the second mutation, or “second hit”, observed in DICER1-related tumors is nearly always limited to the RNase IIIb domain. These somatic second hits are highly-characteristic, affecting amino acid positions 1705, 1709, 1809, 1810 and 1813 that alter but do not destroy DICER1. These ‘hotspots’ are either metal-binding residues, which are important for enzyme function, or lie adjacent to these residues. Mutations in these amino acid positions lead to loss of 5p microRNA synthesis. Loss of heterozygosity (LOH) is uncommon in DICER1-related tumors as a second somatic event, but does occasionally occur, especially in the case of pineoblastoma.
Somatic mosaicism for hotspot mutations in the RNase IIIb domain has been reported in children with severe phenotypes notable for multiple primary tumors, and in some cases, overgrowth. These mutations appear to be accompanied by somatic truncating mutations in the other allele or LOH in the tumors.
Tumor Spectrum
PPB is a rare tumor that arises during fetal and infant lung development. It is thought to be the most common life-threatening manifestation of DICER1 syndrome. From birth until approximately age 2, PPB typically presents as a cystic mass (Type I PPB). From approximately age 2 to age 6, PPB may present as a combination of lung cysts and solid tumor (Type II PPB), or as purely solid tumor (Type III PPB). Types II and III PPB are highly malignant tumors with cure rates that are much lower than in Type I PPB. Type I PPB is capable of progressing to Types II and Types III, emphasizing the importance of early detection and resection. Children with PPB may present with shortness of breath and pneumothorax due to cyst rupture. In cases of advanced disease, they may additionally present with weight loss and fever. PMID: 8636815, 25209242, 19556464.
CN is generally considered a benign renal dysplasia, consisting of multiple thin-walled multilocular cysts. It is one of the more frequent manifestations of DICER1 syndrome, typically occurring below the age of 4 years in mutation carriers, presenting as a painless abdominal or flank mass. While non-neoplastic, partial or total nephrectomy may be required due to a mass effect, or because of impairment of renal function. In very rare cases, CN has been observed to evolve into anaplastic sarcoma of the kidney, a malignant neoplasm. PMID: 21036787, 17137906, 24481001, 28177962
MNG is characterized by nodular enlargement of the thyroid gland. MNG is the most frequent manifestation of DICER1 syndrome, and presents as a palpable neck mass, typically from childhood to early adulthood. MNG and thyroid disease is common in the general population, and therefore DICER1 syndrome should be considered for MNG only in the presence of additional DICER1-related tumors in the individual or family. Differentiated thyroid cancer is a rare manifestation of DICER1 syndrome, and has been observed in children who have been heavily treated (chemotherapy, radiotherapy) for a previous diagnosis of PPB, as well as in those with no prior risk factors for thyroid cancer. PMID: 21205968, 24617712, 27459524.
OSCSTs are a group of uncommon non-epithelial ovarian cancers, and include granulosa cell tumors, gynandroblastomas and Sertoli-Leydig cell tumors (SLCT). SLCT is the most commonly observed OSCST in DICER1 syndrome. While SLCT is a malignant tumor, associated mortality is low. SLCT frequently presents with signs of virilisation (hirsutism, voice changes), along with typical features of an ovarian neoplasm, including abdominal distension and pain. PMID: 21205968, 21501861, 25844550, 27858560.
PitB is an extremely rare primitive tumor of the pituitary gland typically presenting by age 24 months, often with Cushings syndrome, or with opthalmoplegia, and/or hydrocephalus. PitB is a life-threatening tumor. It is a very rare but very highly characteristic manifestation of DICER1 syndrome. PMID: 24839956.
PinB is a rare primitive neuroectodermal tumor of the pineal gland. Presenting features include symptoms of pineal or pituitary mass and ophthalmologic changes. PinB is a rare, yet characteristic manifestation of DICER1 syndrome. It tends to occur at younger ages if a germline DICER1 mutation is present. PMID: 25022261.
ERMS is a frequent pathological finding in tumors related to DICER1 mutation. It is the most prevalent sarcomatous subtype in the mixed-pattern-sarcoma of PPB. ERMS of certain other anatomic sites are highly characteristic in DICER1 syndrome:
ERMS of the uterine cervix (cERMS), also known as “cervical sarcoma botryoides”, is very rare but appears to be highly characteristic of DICER1 mutation. cERMS typically presents between ages 10 to 25 years with abnormal spotting or a polypoid vaginal mass. PMID: 22180160, 24151152, 22157934, 27896549.
Ovarian ERMS (oERMS) has been observed very rarely and may be characteristic of DICER1 mutation. (It is possible that oERMS is not a separate entity but is a subcategory of ovarian SLCT in which ERMS predominates among the heterologous elements found in DICER1-related SLCTs). PMID: 25844550, 25836323.
Bladder ERMS is among the more common ERMS sites in young children, but it appears as if most cases are not related to DICER1 mutation. There are several case reports, with or without germline DICER1 mutations that suggest that bladder ERMS is part of DICER1 syndrome but no cases with somatic “second hit” mutations in DICER1 have been published. PMID: 22180160, 18000857, 20981696.
Vaginal/vulvar ERMS and orbit ERMS (other characteristic early childhood ERMS sites) have not been observed. The alveolar RMS pattern has also not been associated with DICER1 mutations.
NCMH is a very rare benign tumor of the nasal cavities and sinus, often presenting with nasal congestion in childhood or adolescence. NCMH is a rare, but somewhat characteristic manifestation of DICER1 syndrome. PMID: 25118636.
Other tumors/dysplasias that may occur in individuals with germline DICER1 mutations include Wilms tumor, neuroblastoma, medulloblastoma, intestinal juvenile hamatomatous polyps, mesenchymal hamartoma of the liver and liver cysts and atypical paratesticular sarcomas. The vast majority of cases are not caused by DICER1 mutations, and observing any of these features alone in a patient is not sufficient to suggest DICER1 syndrome. However, the risk of these conditions appears to be increased in DICER1 mutation carriers, and genetic testing for DICER1 mutations may be indicated if these are present in the context of additional DICER1-related tumors in the family. Recently, four cases of malignant thyroid gland teratomas and two presacral teratoid neoplasms have been reported as DICER1-associated tumors. DICER1 syndrome was established in one of the patients with a presacral teratoid neoplasm due to availability of germline DNA, the other cases were only somatically evaluated. PMID: 23620094, 28766837, 25190313, 31067372, 32222066, 31917706, 31296931.
Adapted from Foulkes WD, Priest JR, Duchaine TF. DICER1: mutations, microRNAs and mechanisms. Nat Rev Cancer. 2014 Oct;14(10):662-72. doi: 10.1038/nrc3802. Epub 2014 Sep 1.
| wdt_ID | Phenotype and Relative Frequency | Notation | Does this disease by itself suggests DICER1 Mutation Testing? | Approximate Ages of Susceptibility, range (peak). | Malignant or Benign | Deaths recorded in cases with DICER1 syndrome |
|---|---|---|---|---|---|---|
| 3 | Most Frequent Phenotypes | |||||
| 4 | pleuropulmonary blastoma | PPB | yes | |||
| 5 | Type I (cystic) PPB | 0 - 24 m (8 m ) | M | y, if progresses to Type II or III | ||
| 6 | Type II (cystic/solid) PPB | 12 - 60 m (31 m) | M | y, ~40% | ||
| 7 | Type III (solid) PPB | 18 - 72 m (44 m) | M | y, ~60% | ||
| 9 | Type Ir (cystic) PPB | any age | B or M | none observed | ||
| 10 | multinodular goiter | MNG | no | 5 - 40 y (10 - 20 y) | B | n |
| 11 | cystic nephroma | CN | yes | 0 - 48 m (undetermined) | B | n (see anaplastic sarcoma of kidney below) |
| 12 | Sertoli-Leydig cell tumor of ovary | SLCT | yes | 2 - 45 y (10 - 25 y) | M | y, < 5% of cases |
| 13 | Moderate Frequency Phenotypes |
Detection
The identification of DICER1 germline mutations in patients and their family members is important in assessing future neoplasia risk, and allows the opportunity for predictive testing for families to identify individuals at risk for DICER1-related tumors.
Due to the unique and rare nature of many of the DICER1-related tumors, the presence of any of the distinctive tumor types on its own (PPB, CN, SLCT, cERMS, PitB, PinB, NCMH and CBME) would merit a referral to a cancer genetics service for counseling and germline genetic testing of the DICER1. As well, a family history contributory for the presence of less distinctive DICER1-related features, such as early-onset MNG or Wilms tumors, may merit a referral to genetics if they occur in families with DICER1-related tumors in close relatives. Multiple cases of MNGs in a family may be sufficient to consider a referral to a genetics service.
The identification of a germline DICER1 mutation allows for individuals to receive tailored medical follow up and to consider screening for DICER1-related tumors. Tumor surveillance allows for the identification of a tumor at an earlier stage, so that it can be removed and treated with the least possible consequences. Specifically, screening mutation-carrying newborns for early, cystic Type I PPB may allow surgical removal and prevention of the disease progressing to Type II or III PPB which carry much more unfavorable prognoses. However, many DICER1 phenotypes tend not to be life-threatening and occur over an extended age range. Thus, multi-year screening rituals must be weighed carefully against the potential morbidity of such programs.
Unaffected family members of an individual with a germline DICER1 mutation can be tested for the familial DICER1 mutation to confirm or rule out at-risk status. Because the penetrance of DICER1 syndrome for tumors appears to be quite low, cascade genetic screening of relatives may identify the familial DICER1 mutation in unaffected relatives who may be at risk for tumors, or may benefit from the knowledge for early intervention, such as genetic testing and screening for tumors in infants.
Surveillance
There are no established guidelines for tumor surveillance in DICER1 mutation carriers.
The following screening strategies have been suggested by the AACR childhood cancer predisposition workshop (Schultz KAP et al, PTEN, DICER1, FH and their associated tumor susceptibility syndromes: Clinical features, genetics and surveillance recommendations in childhood. Clin Cancer Res 2017 15;23(12):e76-e82). Specific institutions may have different recommendations. It is important to recognise that most of the data are based on expert opinion only.
| Organ | Tumor types observed | Presenting symptoms | Surveillance in known mutation carriers |
| Lungs | PPB Types I, II, III, Ir | Respiratory distress, chest pain, fever and weight loss | – Initial chest CT between 3 to 6 months of age. – If normal, chest CT between 2.5 to 3 years of age. – Chest radiograph should be considered every 6 months up to 8 years of age, and then annually from 8 to 12 years of age. |
| Kidneys | CN, ASK, WT | Abdominal mass, pain, blood in urine
| – Consider abdominal ultrasound every 6 months until age 8. – Annually thereafter until aged 12, depending on the findings. |
| Brain | PinB, PitB | Pineoblastoma: symptoms of pineal or pituitary mass, ophthalmologic changes. Pituitary blastoma: Cushing syndrome, ophthalmoplegia, or diabetes insipidus. | – Surveillance brain MRI may be considered but is controversial, as risk/benefit ratio is not yet known, and these tumors are both rare manifestations of DICER1 syndrome (<1%). – Urgent brain MRI if any symptoms of an intracranial pathology are present. |
| Gynecologic | OSCST (SLCT, gynandroblastoma), cERMS, oERMS | OSCST: abdominal mass/distension, menstrual irregularities, signs of virilisation. cERMS: vaginal spotting, polypoid mass extending from vagina. | – Consider annual or semiannual pelvic ultrasound (at the same time as renal surveillance) every 6 to 12 months from birth into adulthood. |
| Thyroid | Thyroid nodules, MNG, differentiated thyroid cancer | Palpable nodules, local compressive symptoms (dysphagia, dysphonia, stridor) | – Consider thyroid ultrasound with assessment for local adenopathy at age 8 years. – If normal, repeat every 3 years. |
| Eye | CBME | Unusual appearance to pupil, vision impairment | – Awareness of risk. |
| Nose | NCMH | Nasal congestion or “stuffiness” | – Awareness of risk. – ENT with nasal endoscopy if symptoms of nasal obstruction are present. |
| GI | Hamartomatous polyps | Intestinal obstruction – bloating, pain | – Awareness of risk. – Evaluation if symptoms of intestinal obstruction are present. |
* These recommendations may change as more data is available regarding the spectrum of tumors in DICER1 syndrome.
There is no evidence to suggest that tumors associated with germline DICER1 mutations have different treatment outcomes than the same tumors occurring sporadically.
Most tumors in the DICER1 syndrome spectrum have a relatively benign course. However, malignancy does occur in certain DICER1-related tumor types, notably PPB. Others are associated with morbidity and/or mortality due to location and growth effects, such as PitB, or clinical manifestations such as androgenisation due to SLCTs. Because DICER1 syndrome is rare, physicians encountering DICER1-related diseases should consider contacting syndrome experts for latest treatment recommendations.

